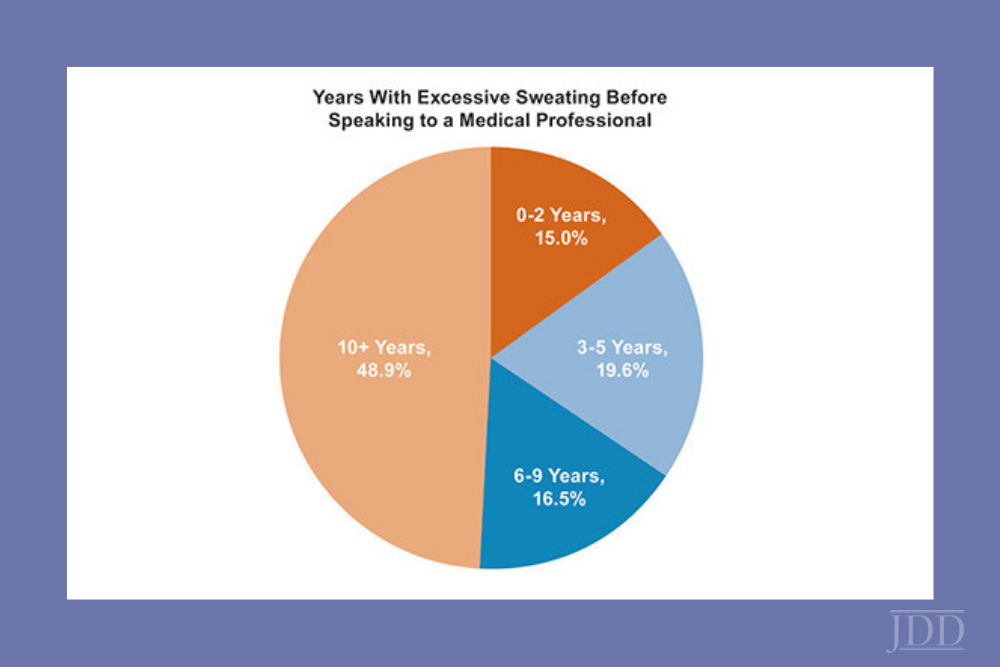
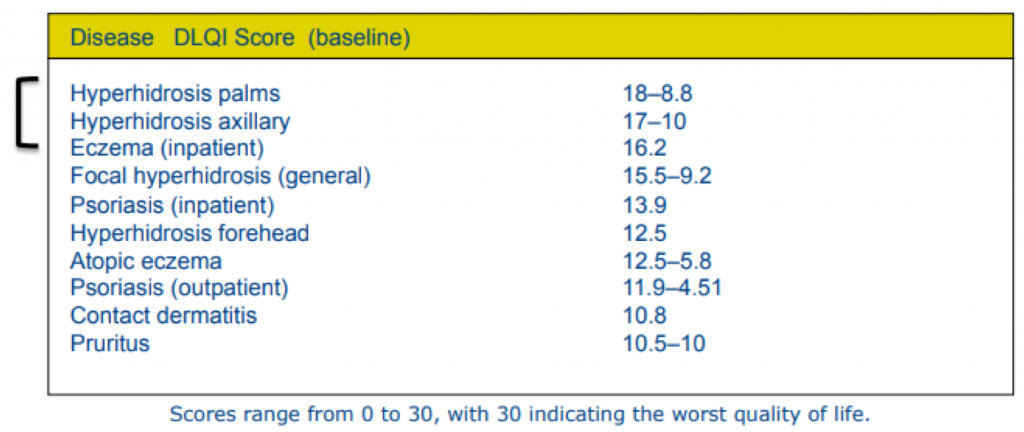
Hyperhidrosis affects nearly 400 million people worldwide, yet it’s often misunderstood and underdiagnosed. In fact, 27% of adults never receive a diagnosis, according to the International Hyperhidrosis Society. The condition greatly impacts a person’s quality of life: People with excessive sweating face nearly three times the risk of anxiety and depression.
November is Hyperhidrosis Awareness Month. Let’s bring excessive sweating out of hiding and help people find understanding and treatment.
Join us for the ODAC Pre-Conference Virtual Symposium on Tuesday, November 11, where dermatologist Dr. Angela Lamb will share strategies and new tools to improve quality of life in patients with excessive sweating. You’ll also experience case-based sessions on chronic itch, hidradenitis suppurativa, and pediatric alopecia areata. Registration is complimentary – plus you can earn CME!
We hope to see you at this interactive event that’s sure to provide you with practical takeaways you can immediately apply in clinic.
Improve your knowledge and provide hyperhidrosis patients with hope and healing.