ODAC Dermatology Conference, in partnership with Next Steps in Derm, interviewed Dr. David Miller, Instructor in Dermatology and Medicine at Harvard Medical School and member of the Department of Dermatology and the Department of Medicine at Massachusetts General Hospital, where he is co-director of the Merkel cell carcinoma treatment program. Watch as he shares important updates on Immuno-Oncology (IO) strategies for melanoma.
Next Steps in Derm, in partnership with ODAC Dermatology, Aesthetic and Surgical Conference, interviewed Dr. Vic Ross, Director of the Scripps Clinic Laser and Cosmetic Dermatology Center in San Diego, CA, on his approach and the various interventions he uses for the treatment of Melasma. Watch as he shares fresh practical pearls immediately useful in your practice.
So many diseases, so few FDA approved indications. Between the mind-numbing time and cost from bench to bottle, it is no surprise that dermatologists, as masters of the integument are the off-label bandits, marrying their wealth of knowledge on the pathophysiology of skin diseases to the mechanisms by which medications work to create an evidenced-based armament of therapies for both common and rare diseases alike. Where a primary care physician sees a blood pressure medication, a dermatologist sees an effective medication for acne. A medication for malaria you say? They use it for a plethora of complex diseases ranging from Lupus to scarring hair loss. The lengthy list of medications used off-label in dermatology continues to grow and for good reason and fortune.
Next Steps in Derm, in partnership with ODAC Dermatology, Aesthetic and Surgical Conference interviewed Dr. Adam Friedman, Professor, Interim Chair of Dermatology, and Residency Program Director at George Washington University, who shared off-label uses of drugs such as Hydroxychloroquine (currently under investigation for the treatment of patients with COVID-19), Pentoxifylline, and Colchicine. A wealth of clinical pearls you don’t want to miss!
Additional video pearls can be found here.

Source: Journal of Drugs in Dermatology
While the world lives under the shadow of the novel coronavirus (COVID-19) pandemic, dermatologists wonder if the current situation calls for a temporary change in the management of skin conditions.
Immunosuppressive drugs are used ubiquitously in the modern treatment of inflammatory and autoimmune skin diseases like psoriasis, bullous diseases, connective tissue diseases, and many others. Treatment of these conditions is based on the suppression of the patient’s immune system using steroids, steroid-sparing drugs, and biological agents.1
While the effects of the novel coronavirus on the body and its immune system are still being studied, there is overwhelming evidence that the virus could directly or indirectly affect the immune system. In one study, lymphocytopenia was reported in 83.2% of the admitted patients and might be associated with a worse prognosis.2In another study, a steady decline in the lymphocyte counts was recorded in a group of patients who did not survive the infection.3
While the coexisting comorbid medical conditions (such as diabetes or heart disease) are considered as independent predictors of an adverse outcome of the novel coronavirus infection,4 it could be assumed that the chronic inflammatory and autoimmune skin diseases like psoriasis by themselves might imply an additional risk factor of developing more serious symptoms of the novel virus due to their chronicity and effects on the immune system.5
The use of immunosuppressive to treat these conditions can amplify this effect, and it might leave the patient vulnerable to more serious complications should an infection with the novel coronavirus be established. Hence, it may be wise to restrict temporarily the use of immunosuppressive agents including systemic steroids, steroid-sparing agents, and biologics in dermatology daily practice until more evidence is available about their safety in the current pandemic.6As a relates point, the International Psoriasis Council declared an urgent statement on March 11, 2020 that the physician should be alert to the potentially harmful effects of COVID-19 infection on patients with psoriasis and to immediately discontinue or postpone immunosuppressant medications for psoriasis patients diagnosed with COVID-19 disease.7
Clinical photography is a critical tool for the dermatologist and has rapidly become standard of care in the digital era. While a point-and-click approach to photography is sufficient for some circumstances, there are some simple tricks and techniques that will elevate your photography to a new level of professionalism.
Next Steps in Derm, in partnership with ODAC Dermatology, Aesthetic and Surgical Conference interviewed Dr. Justin Finch, Associate Professor of Dermatology at UConn and Co-Founder of Central Connecticut Dermatology who shared his top 3 pearls for improving clinical photography in your practice.
Source: ODAC Dermatology, Aesthetic & Surgical Conference (ODAC) Discovery in Dermatology Poster Session
At the 17th Annual ODAC Dermatology, Aesthetic, and Surgical Conference (ODAC) held January 17-20 in Orlando, FL, Brandon Kirsch, MD, Janet DuBois, MD, Martin N. Zaiac, MD and Deepak Chadha, MS, MBA, RAC presented scientific research of long term data with a novel therapeutic for topical treatment of primary axillary hyperhidrosis in pediatric subjects.
Discovery in Dermatology
The use of retro-metabolically designed drugs in dermatology is novel and has the potential for providing significant therapeutic benefit to pediatric and adult patients.
Sofpironium bromide is an ester analogue of glycopyrrolate that inhibits muscarinic receptors in sweat glands. It was developed according to the principles of retro-metabolic drug design, in which the goal is to create an active compound that is metabolized in vivo to an inactive moiety in a single, predictable reaction. Retro-metabolically designed drugs are rapidly metabolized in the bloodstream, potentially allowing for optimal therapeutic effect at application sites with minimal systemic side effects.
Introduction
~2.1% of the US population aged <18 years has primary hyperhidrosis (HH); ~65% have axillary HH. Long-term safety/tolerability and efficacy of topical HH treatments have rarely been studied in pediatric patients. Sofpironium bromide is a retro-metabolically designed analog of glycopyrrolate (anticholinergic) in development for topical treatment of primary axillary HH. Absorbed drug is rapidly metabolized, potentially allowing optimal local therapeutic effect with minimal systemic effects..
Procedures
21 of 25 subjects (age 9-16 yrs) with primary axillary HH of ≥6 months duration, completing a previous 1-week safety and pharmacokinetic (PK) study (BBI-4000-CL-105), were enrolled. Objectives were to assess safety/tolerability and PK, and explore efficacy of sofpironium bromide gel, 15% applied to both axillae for 24 weeks.
Results
Mean age (SD) 13.3 (2.29) years. 16 subjects completed this 24-week study. 7 had treatment emergent adverse events (TEAEs); 4 with AEs related to study drug, including expected systemic anticholinergic AEs (blurred vision, dry mouth, dry eyes, mydriasis) and local events (pain, pruritus, rash, erythema). 2 subjects discontinued due to TEAEs, including dry eye, dry mouth, local pruritus, local rash. The majority (52.4%) of subjects did not have any local symptoms/signs, and none observed were severe in nature. PK did not show evidence of drug/major metabolite accumulation, with most subjects having concentrations not quantifiable. The validated patient-reported outcome, Hyperhidrosis Disease Severity Measure-Axillary (HDSM-Ax), showed mean (SD) change from baseline (from previous study) to Week 24 of this study of -1.91 (1.038). A -1.00 change shows clinically meaningful improvement.
Conclusion
In this 24-week study in pediatric subjects sofpironium bromide, 15% was safe/well tolerated. Majority of subjects had no TEAE, and there were no severe or serious AEs. There was no evidence of drug accumulation. There was indication of clinically meaningful improvement in axillary HH.
During the 2020 ODAC Dermatology, Aesthetic and Surgical Conference, Dr. Amy McMichael, Professor and Chair of Dermatology at the Wake Forest University School of Medicine, sat down with Next Steps in Derm to share important updates regarding treatments on the horizon for the most common forms of hair loss. Dr. McMichael will be presenting at Skin of Color Update 2020 with lectures including Hair & Scalp Disorders in SOC: Diagnostic Approaches and Hot Topics & Controversies in Photoprotection: Making sense of it all.
Source: Next Steps in Dermatology
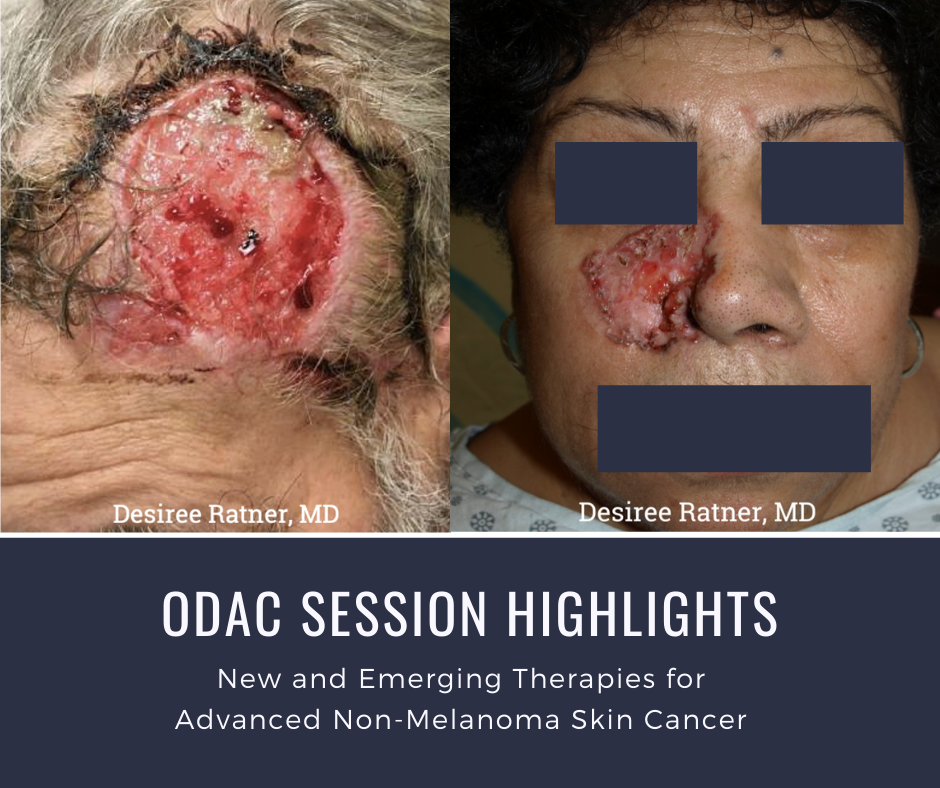
At the 17th Annual ODAC Dermatology, Aesthetic, and Surgical Conference (ODAC) held January 17-20 in Orlando, FL, Dr. Desiree Ratner led a discussion on new and emerging therapies for advanced non-melanoma skin cancer discussion.
Treatment Options
The session covered several treatments for patients including patidegib gel 2% and 4% applied once or twice daily in patients with basal cell carcinoma. Patidegib is a topical hedgehog inhibitor made by PellePharm and its mechanism of action is to block Smo signaling, thereby inhibiting the hedgehog pathway that contributes to the development of basal cell carcinomas. This treatment has several advantages in that it does not contribute to hair loss, taste loss, or muscle cramps. It has the potential to treat and mitigate facial basal cell carcinomas in basal cell nevus patients. It is being studied in randomized clinical trials enrolling patients with Gorlin’s syndrome (basal cell nevus syndrome) in the United States and in Europe.
Hedgehog pathway inhibitor resistance is unusual but may occur as “rebound” tumor growth after drug cessation or secondarily after long-term smoothened inhibitor therapy. Resistance to hedgehog pathway inhibitors is classified into primary and secondary resistance. Primary resistance has been postulated to bypass mechanisms of genes downstream of smoothened, such as the G497 W mutation. Secondary resistance in patients who showed an initial response has actually been thought to be due to de novo mutations located on regions in smoothened to which hedgehog pathway inhibitors bind or selective clonal expansion of minority clones in the pre-treated tumor. Further studies are definitely needed to elucidate what drives resistance to hedgehog pathway inhibitors and how basal cell carcinoma resistance may be overcome by other novel, emerging therapies.
Patient Cases
Dr. Ratner presented a number of interesting patient cases with advanced basal cell carcinomas sometimes so large that patients lose mobility and function of a body part or organ. In most cases, locally advanced BCCs respond well to oral hedgehog inhibitors, which can be used for long-term control or neoadjuvantly prior to surgery. In the case of one patient, an aggressive orbital BCC caused contraction of the tissues around his eye, such that he was not able to open it. Despite treatment with an oral hedgehog inhibitor, his tumor continued to grow, resulting in destruction of his orbit and locoregional metastasis.
Samples of his tumor and normal skin were sent to Stanford University, which performed whole exome sequencing. In the studies of these samples, it became evident that the tumor should have responded to vismodegib but had developed resistance due to another as yet unknown mechanism. Therapies designed to override resistance such as second-generation smoothened inhibitors are under development.

Source: Dermatology News
The following is an excerpt from Dermatology News Expert Analysis, Conference Coverage from ODAC.
ORLANDO – Dermatologists should be well versed in addressing common concerns that patients, family members, and the media have about photoprotection, Adam Friedman, MD, advised at the ODAC Dermatology, Aesthetic, & Surgical Conference.
“Know the controversies. Be armed and ready when these patients come to your office with questions,” Dr. Friedman, professor and interim chair of dermatology at George Washington University, Washington, said in an interview at the meeting, where he presented on issues related to photoprotection.
Which SPF to choose and the impact of sunscreen on vitamin D are among the issues patients may be asking about. Sunscreen SPFs above 50 don’t technically provide a “meaningful” increase in ultraviolet protection, given that this value relates to filtering about 98% of UVB, but they still can provide some benefit, which has to do with real-world human error, Dr. Friedman said.

Source: Dermatology News
The following is an excerpt from Dermatology News Expert Analysis, Conference Coverage from ODAC.
ORLANDO – Because Food and Drug Administration–approved treatment options for children and adolescents with severe dermatologic diseases are limited, systemic therapies for these patients often require the use of off-label medications. However, this scenario is changing, A. Yasmine Kirkorian, MD, said at the ODAC Dermatology, Aesthetic & Surgical Conference.
“I really would like to emphasize that children with severe disease need to be treated,” added Dr. Kirkorian, a pediatric dermatologist at George Washington University, Washington, and Children’s National Health System, where she is interim chief of the division of dermatology.
Current on-label systemic therapies for pediatric skin disease include etanercept for psoriasis (4 years and older), ustekinumab for psoriasis (12 years and older), adalimumab for hidradenitis suppurativa (12 years and older), and omalizumab for chronic idiopathic urticaria (12 years and older). A new addition to the list is dupilumab, which was approved for children and adolescents with atopic dermatitis (AD) aged 12 years and older in 2019, she noted.

